UVM Proctor Page
Cloudiness’ affect on refractometer measurements
Refractometers are another tool that can be used to make precise density measurements
By MARK ISSELHARDT AND TIMOTHY PERKINS |
Accurately measuring density is critical to the production of pure maple syrup. Historically, sugarmakers have relied on thermometers and or hydrometers to determine when syrup has reached the desired density. These instruments remain valuable tools today. Refractometers are another tool that can be used to make precise density measurements. Like all instruments, to get accurate measurements they must be used properly and in accordance with the manufacturer’s instructions.
Refractometers (both analog and digital) measure the amount of light refracted (bent) through a given liquid sample in order to determine density. The more dissolved solids, primarily sugar in the maple industry, that are contained in the liquid, the more the light is bent. Digital refractometers possess their own light source and light sensor whereas analog refractometers rely on sunlight as the light source and the readers eye as the sensor. Analog refractometers also require the user to visually interpret where the “shadowline” crosses the graduated scale. Digital refractometers have an LCD display that automatically provides the user with the density value. Several companies market refractometers (digital and analog) directly to the maple industry.
The accuracy of a refractometer is greatly impacted by temperature. The apparent density of a liquid decreases with increasing temperature. It is important therefore that the temperature of the sample, temperature of the instrument and the air temperature can all be in equilibrium before a measurement is taken. Some manual and most digital refractometers feature automatic temperature compensation (ATC) for the temperature of the sample being measured. The small mass of the sample used in a refractometer means that hot samples cool dramatically within the first minute of collection. Make sure you understand the temperature range that your instrument is designed to handle.
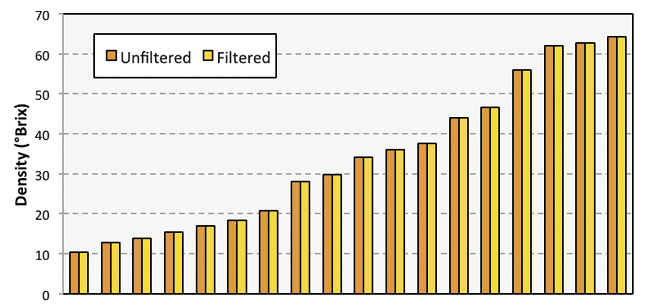
As sap is transformed into maple syrup within an evaporator, it frequently becomes cloudy due in part to mineral precipitates (sugar sand or nitre) and other material that accumulate during the boiling process. The extent of the cloudiness can vary considerably from season-to-season as well as within a season. In addition, many sugarmakers use diatomaceous earth (DE) as a filter aid. Syrup mixed with DE can also appear cloudy. Given the critical role light plays in how a refractometer works, questions often arise as to whether accurate readings with refractometers are possible with cloudy samples. To answer this question, a series of paired filtered and unfiltered concentrated sap samples and syrup samples mixed with DE were measured for density using a digital refractometer.
Eighteen samples (500ml) of partially boiled sap were collected simultaneously from each of six partitions of an oil-fired, 3’X10’ evaporator. The samples captured the entire gradient of density from where sap enters the evaporator to where nearly finished syrup exits the draw-off partition. Samples were placed in clean glass jars and frozen for future analysis. In the lab, samples were allowed to thaw and reach a stable room temperature (~72 °F). A magnetic stirring hot plate (heating element turned off) was used to ensure the samples were well mixed. Temperature of each sample was measured and recorded using a thermocouple probe and data logger (Graphtec model #GL200, Graphtec America). A small subsample of the unfiltered material was then taken and measured for density using a digital refractometer (Palm Abbe, Misco model # PA202, Cleveland, OH). The refractometer has a stated precision of ±0.1 Brix and automatically temperature compensation within a range of 32-167 °F. A subsample was taken and then filtered using a glass fiber syringe prefilter (Fischer Scientific # 0974048C). Density was again measured using the digital refractometer.
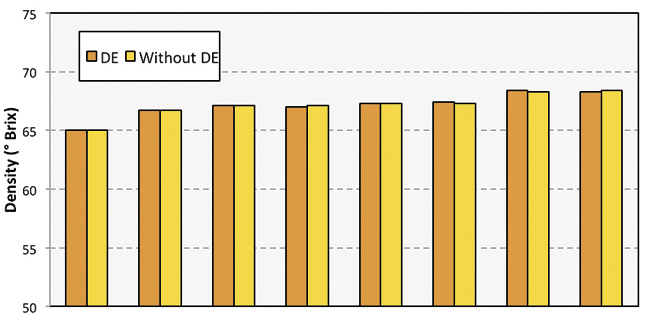
A second experiment used eight samples of pure maple syrup to determine if DE mixed with syrup would yield different refractometer measurements than the pure syrup alone. A subsample of syrup (approximately 20 g) was mixed with 0.20 g DE (Dicalite Minerals Corp.) to produce a visually cloudy sample. Syrup density was measured for each pair (with and without DE) using a digital refractometer.
Filtering was successful at removing visible suspended material (Figure 1). As expected, there was a broad range of density in samples taken throughout the evaporator. Samples near the inlet of the evaporator had a density between 10 to 15 °brix. Samples taken from the drawoff partition were as high as 64.3 brix. Cloudiness caused by niter did not have a detectible effect on refractometric density measurements of any concentrated sap samples (Figure 2).
Syrup mixed with DE appeared visually cloudy (Figure 3). The greatest difference in density measured with a refractometer was 0.1 °brix, which is within the stated precision of the refractometer.
The results of the second experiment demonstrate that digital refractometers measurement of °brix in pure maple syrup and syrup mixed with DE yield the same value (Figure 4). How can it be that a device that relies on light passing thought a liquid would not be affected by a cloudy sample? The construction and operation of a refractometer holds the answer. A refractometer measures the degree to which light is bent (or refracted) while passing through a given sample, but not the amount of light that passes through.
Instruments that measure light transmittance (%Tc) to determine color grade of maple syrup on the other hand are highly impacted by cloudy samples. In a digital refractometer, light source and sensor are on the same side of the sample and not on opposite sides as might be assumed. Suspended material (minerals, DE etc.) therefore does not interfere with the refractive measurement (unless the sample is so cloudy that no light can pass). While a cloudy sample might impact the overall brightness of the image in the eyepiece of an analog refractometer, it does not affect the underlying reading.
It is important to remember several key factors that will help to ensure accurate measurements with a refractometer. Refractometers must be calibrated often (with distilled water or permeate) or measurements can be incorrect. Samples (especially hot samples) placed uncovered on the refractometer will continue to evaporate and can cause irregularly high density measurement. The greatest source of error is likely failing to account for sample temperature. Accurate measurements are obtainable by knowing the limitations of the unit you are using and following the manufacturer’s guidelines carefully.
Aug./Sept. 2014